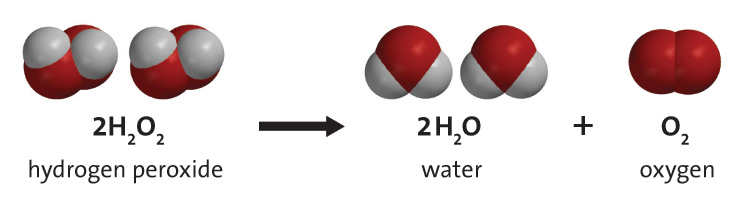
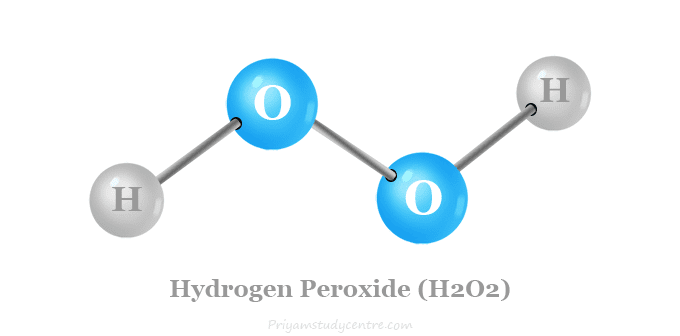
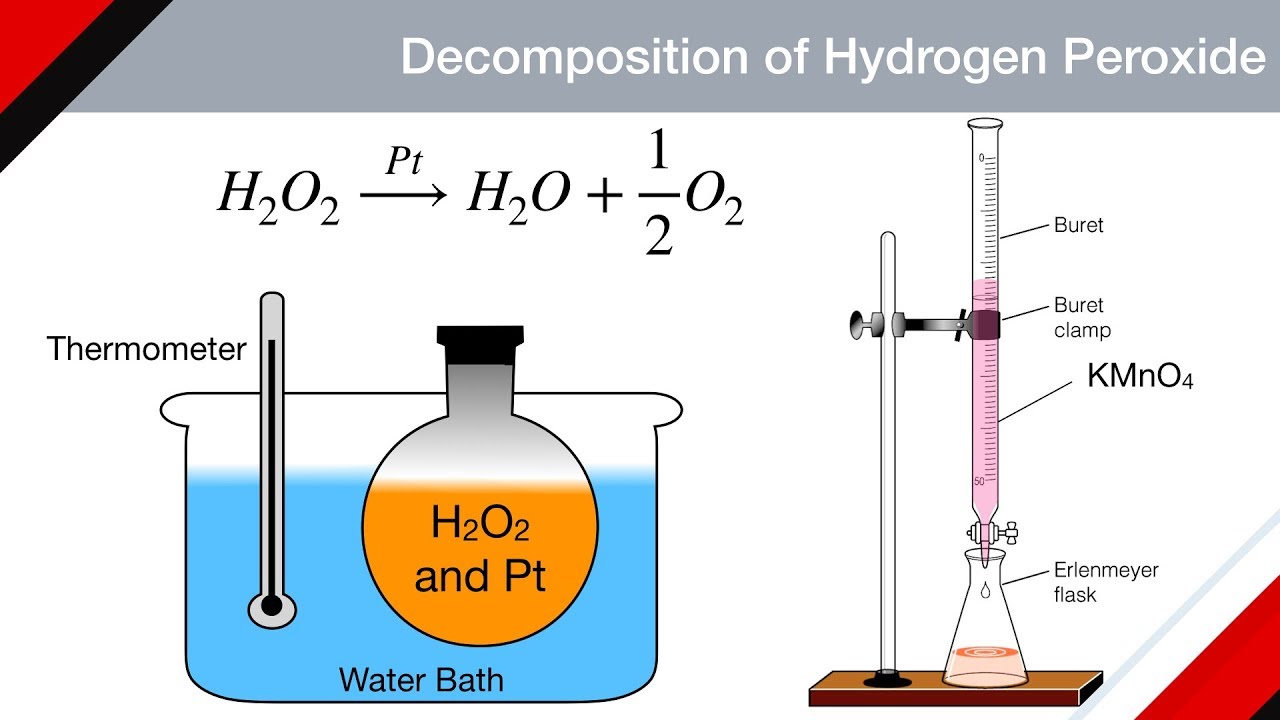
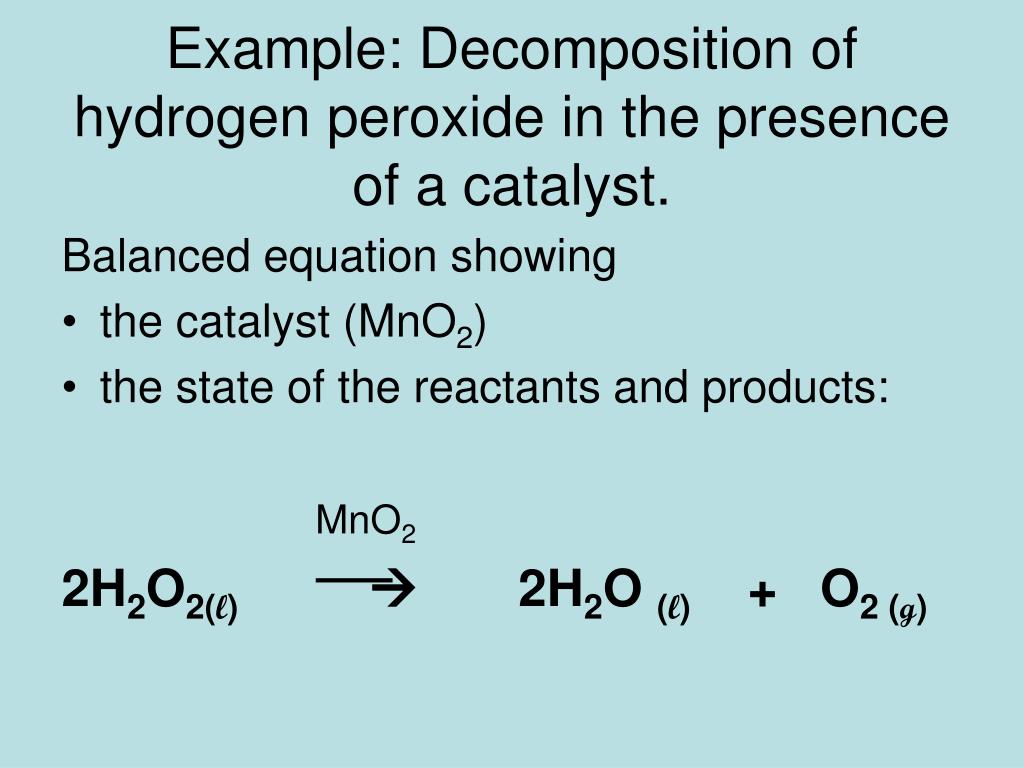
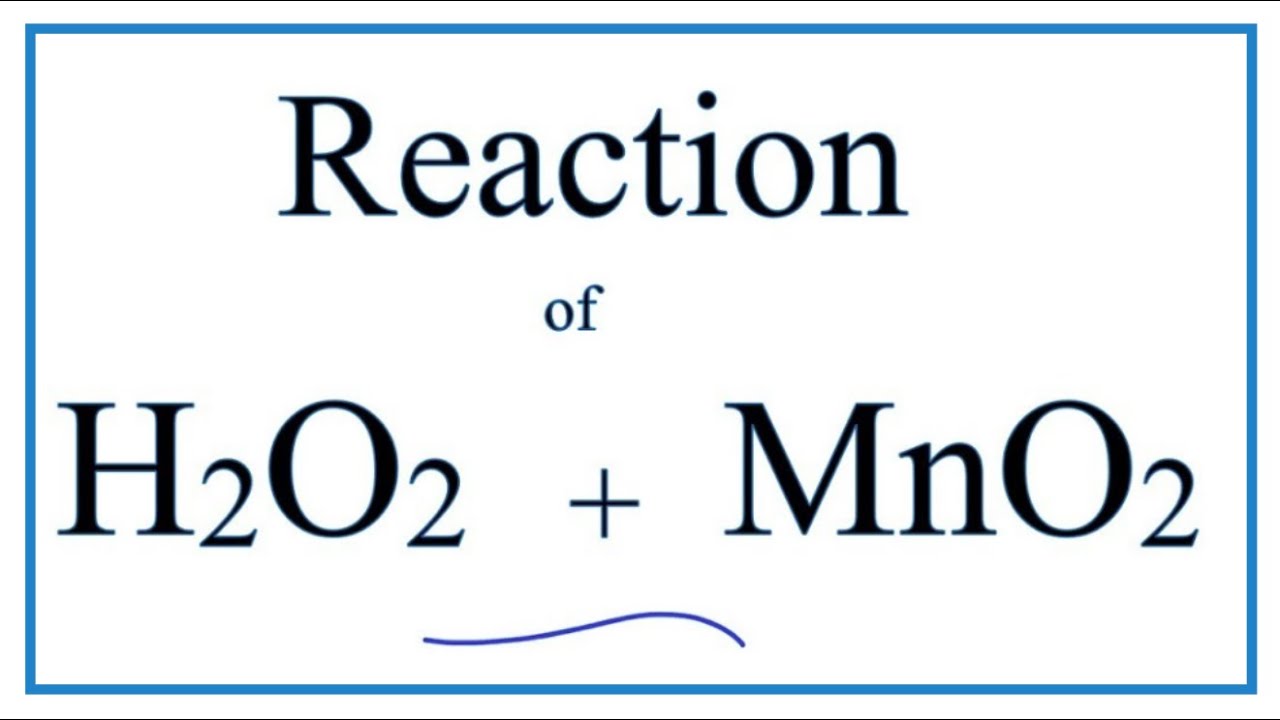OneClass: A proposed mechanism for the decomposition of hydrogen peroxide consists of three elementar...

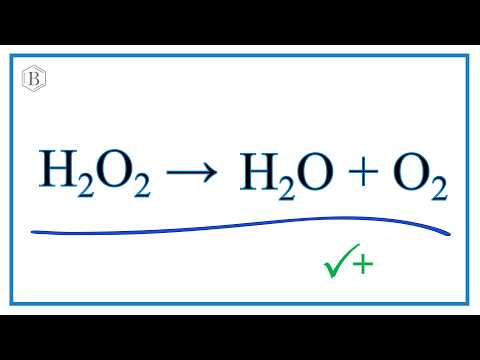
Given below is the chemical equation of decomposition of H2O2. 2H2O2 rarr 2H2O + O2 a) Which catalyst is used for decomposition of H2O2.? b) Which substrate will reamain in the test

Question Video: Using Word Equations to Describe the Decomposition of Hydrogen Peroxide (H2O2) | Nagwa

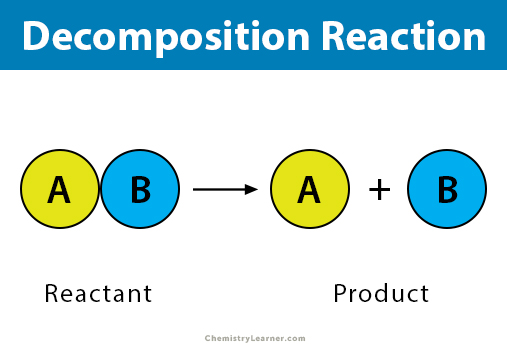
ENTRY QUIZ 1 1.What is chemical reaction? 2.Give an example? 3.What is at the left? 4.What is at the right? 5.What the arrow means? - ppt download

SOLVED:A possible mechanism for the decomposition of hydrogen peroxide is H2O2⟶ 2 OH H2O2+OH⟶H2O+HO2 HO2+OH⟶H2O+O2 Using your results from Exercise 39, specify which step is the rate-determining step. What is the overall

Question Video: Identifying the Balanced Chemical Equation for the Decomposition of Hydrogen Peroxide | Nagwa
![PDF] Decomposition of hydrogen peroxide - kinetics and review of chosen catalysts | Semantic Scholar PDF] Decomposition of hydrogen peroxide - kinetics and review of chosen catalysts | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f2948b1ccb4f9f878523a741f22c7a299fa747b2/2-Figure1-1.png)
PDF] Decomposition of hydrogen peroxide - kinetics and review of chosen catalysts | Semantic Scholar