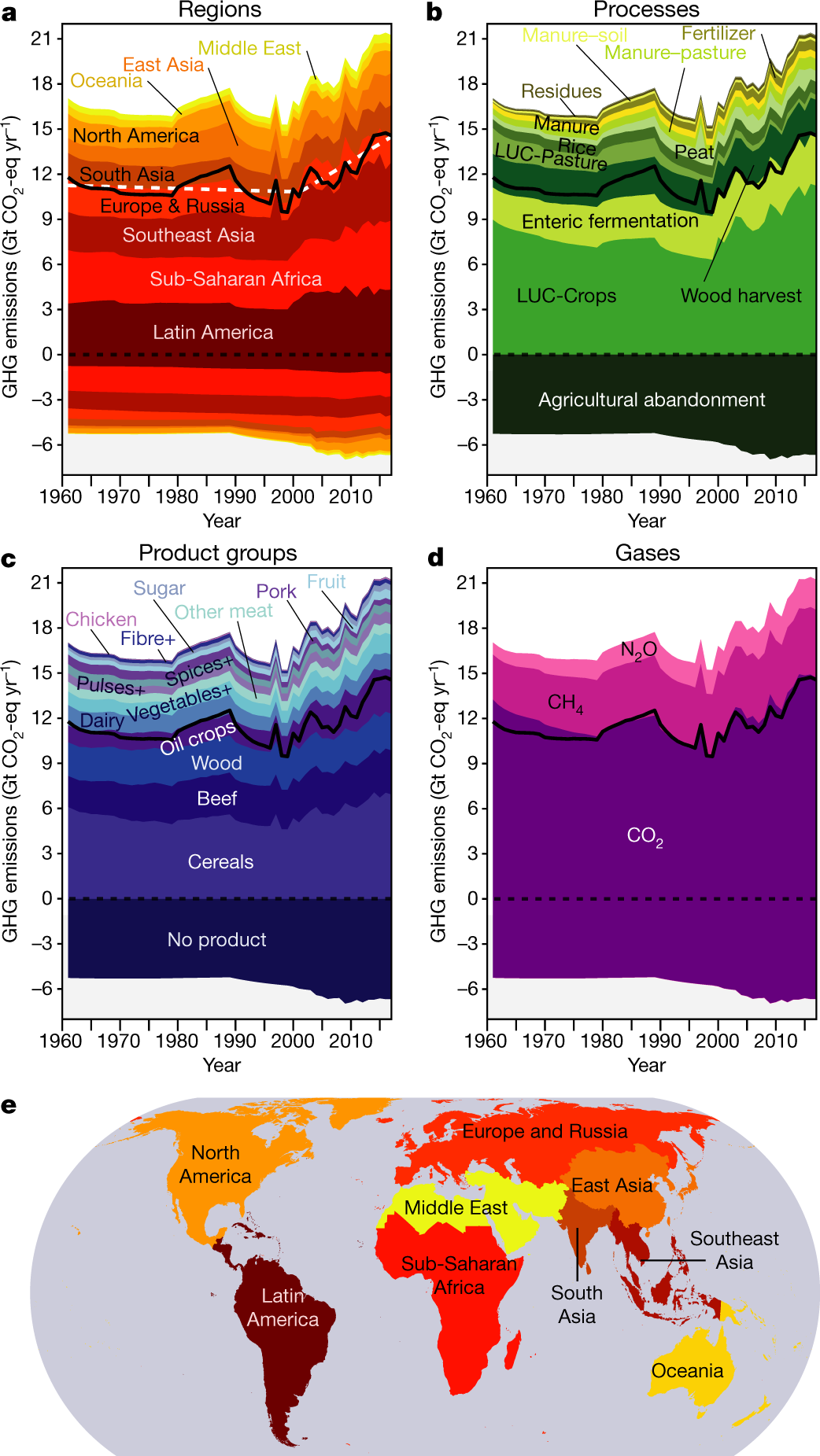
Metal–CO2 Electrochemistry: From CO2 Recycling to Energy Storage - Wang - 2021 - Advanced Energy Materials - Wiley Online Library

8g oxygen, 14 g nitrogen and 22 g carbon dioxide are mixed in a container of volume 4 l. Find out the pressure of the gas mixture at 27^(@)C. Given R =
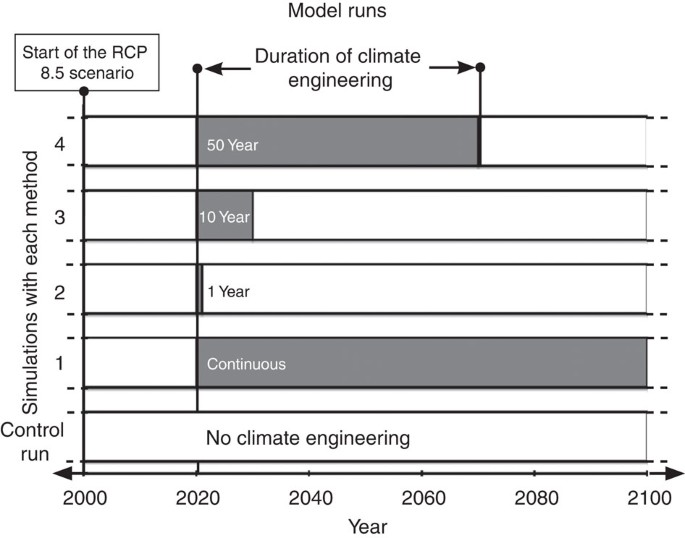
Potential climate engineering effectiveness and side effects during a high carbon dioxide-emission scenario | Nature Communications

8g of oxygen, 14 g of nitrogen and 22 g carbon dioxide are mixed in an encloser of volume 10 litre and temperature 27^(@)C. Calculate the pressure exerted by the mixture ,
A 5.0L sample of oxygen at a pressure of 100kPa and 2.0L sample of nitrogen at a pressure of 250kPa are introduced into a 2.5L vessel. What is the total pressure of

Calculate the work done during isothermal reversible expansion of one mole ideal gas from 10 atm to 1 atm at 300 K.

Methoxy Groups Increase Reactivity of Bifunctional Tetraarylphosphonium Salt Catalysts for Carbon Dioxide Fixation: A Mechanistic Study | The Journal of Organic Chemistry
How to calculate the work done if three moles of an ideal gas are compressed reversibly and isothermally from a volume of 10 litres to 5 litres - Quora

Convert 22 g of carbon dioxide `(CO_(2))` into moles. (Atomic masses : `C = 12 u, O = 16 u`) - YouTube

Calculate the amount of carbon dioxide that could be produced when (i) 1 mole of carbon is burnt in air.(ii) 1 mole of carbon is burnt in 16 g of dioxygen.(iii) 2

Calculate work done when 2 mole of an ideal gas expands isothermally and reversibly at 300 K from 10 atm pressure to 2 atm pressure.

Mechanically Constrained Catalytic Mn(CO)3Br Single Sites in a Two-Dimensional Covalent Organic Framework for CO2 Electroreduction in H2O | ACS Catalysis