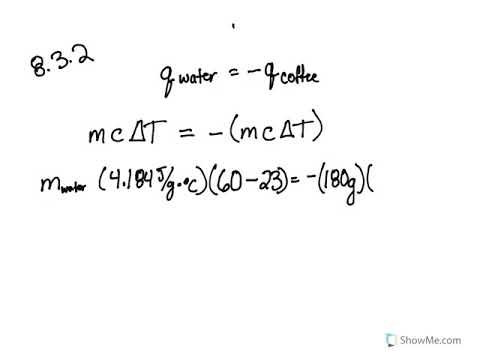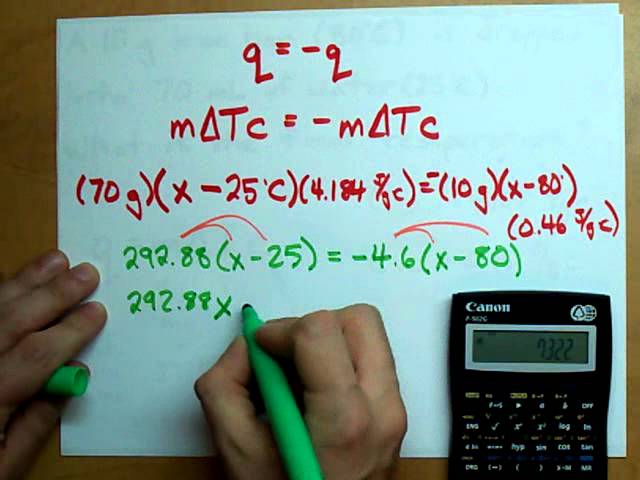What is Specific Heat Capacity? | How to Find Specific Heat Capacity - Video & Lesson Transcript | Study.com

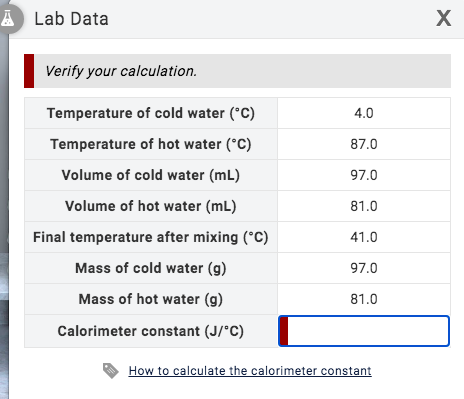
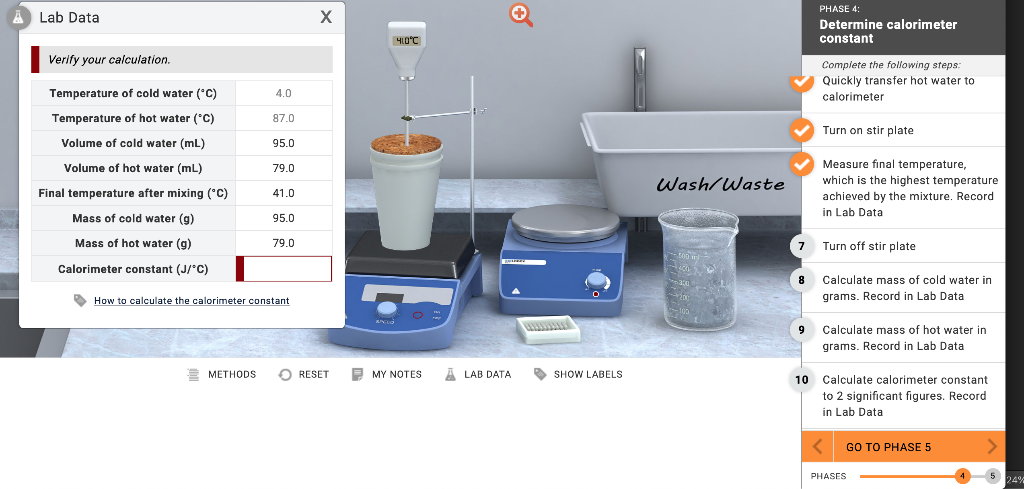
SOLVED: Results: Calculate the heat gained by the water (lost by the substance in the calorimeter using the equation in the Introduction: Remember t0 write the unitsl! Q fon water using Ist