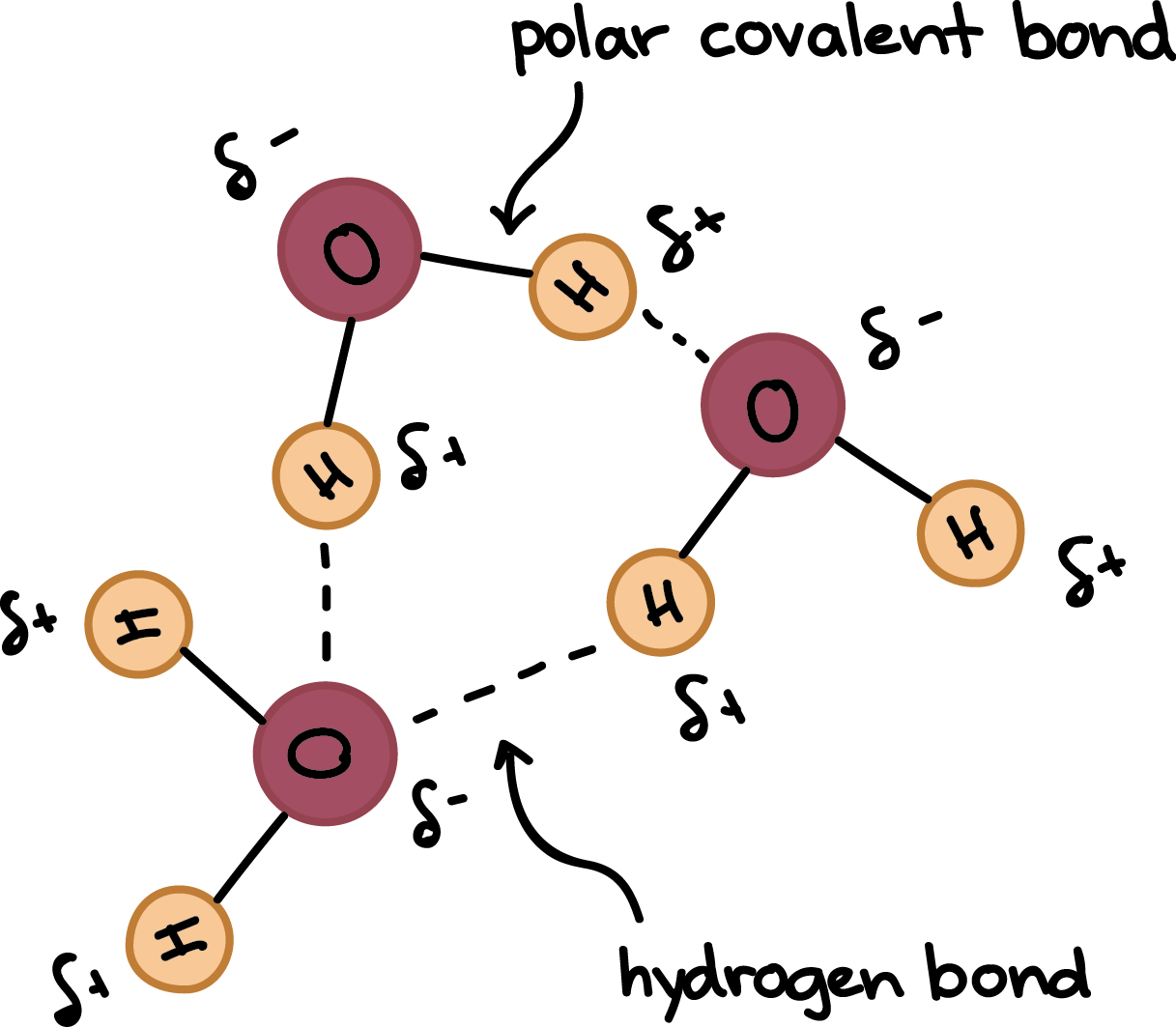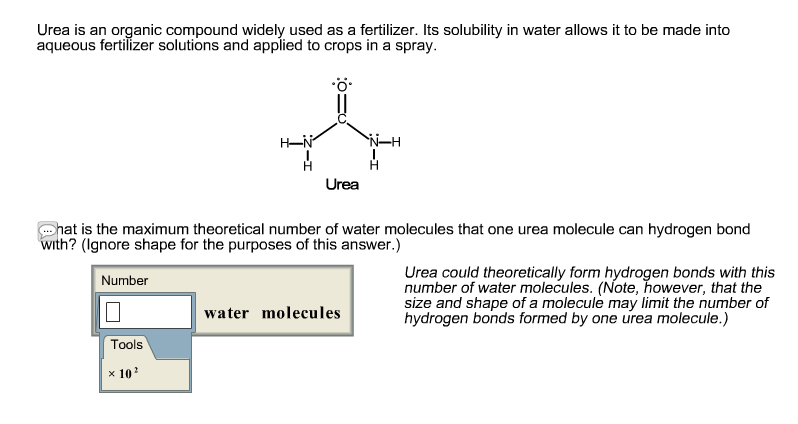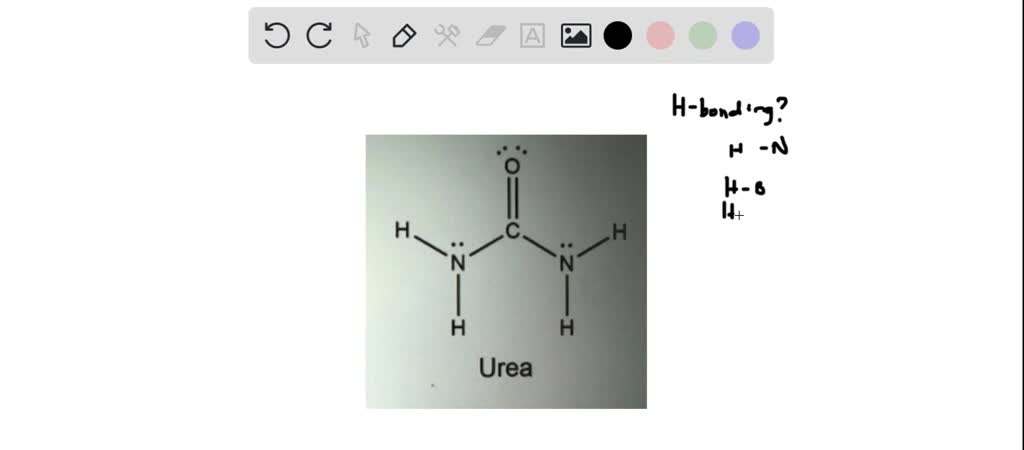
SOLVED: Urea is an organic compound widely used as fertilizer: Its solubility in water allows it to be made into aqueous fertilizer solutions and applied t0 crops in spray: What is the

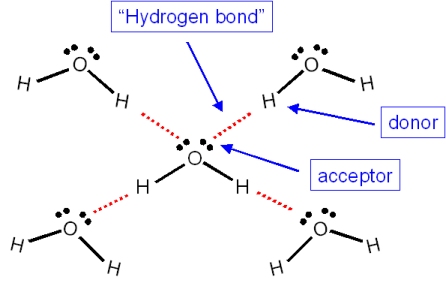
How many hydrogen bonds are attached to each water molecule in a solid state? How many in a liquid state? - Quora

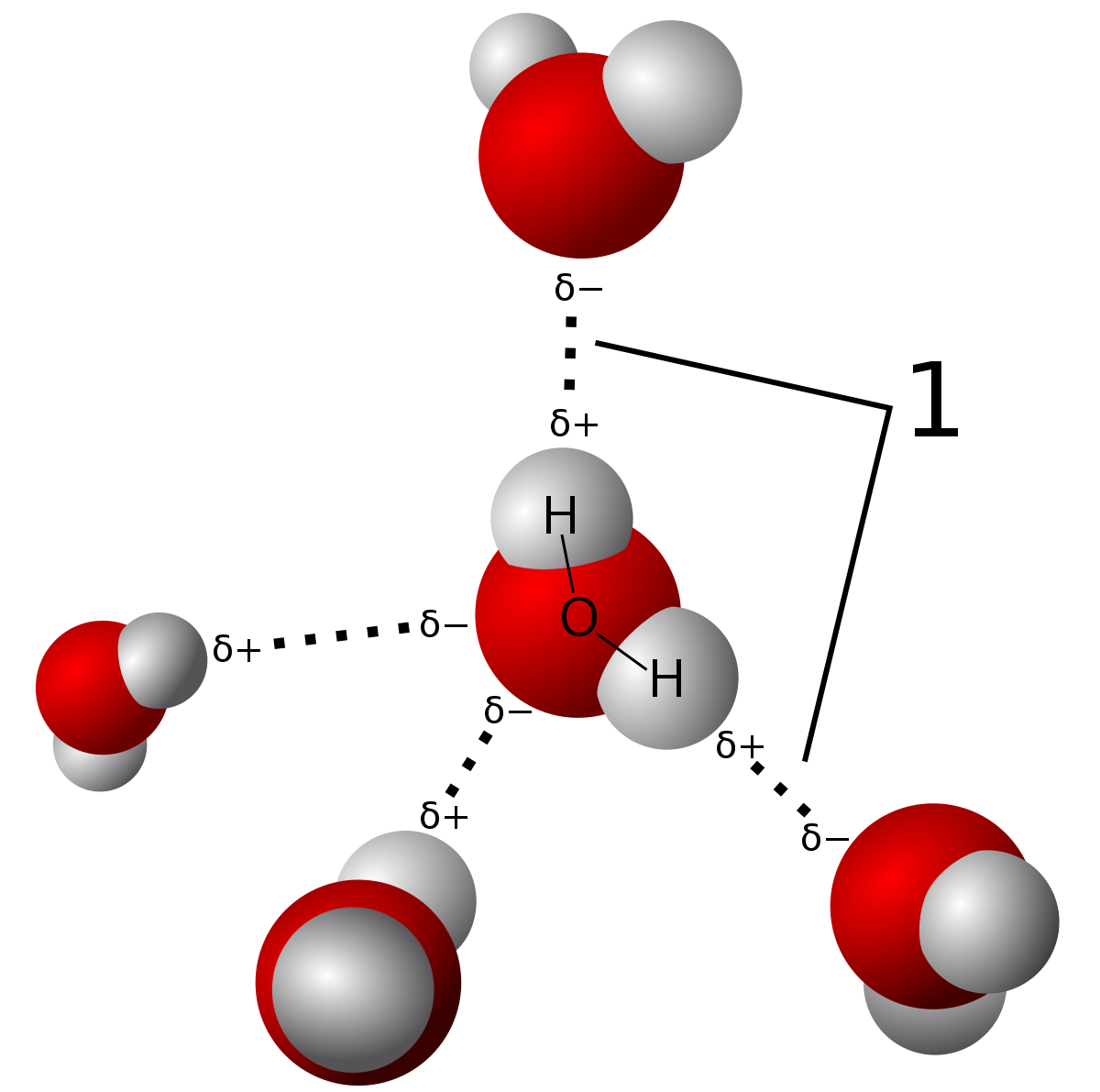
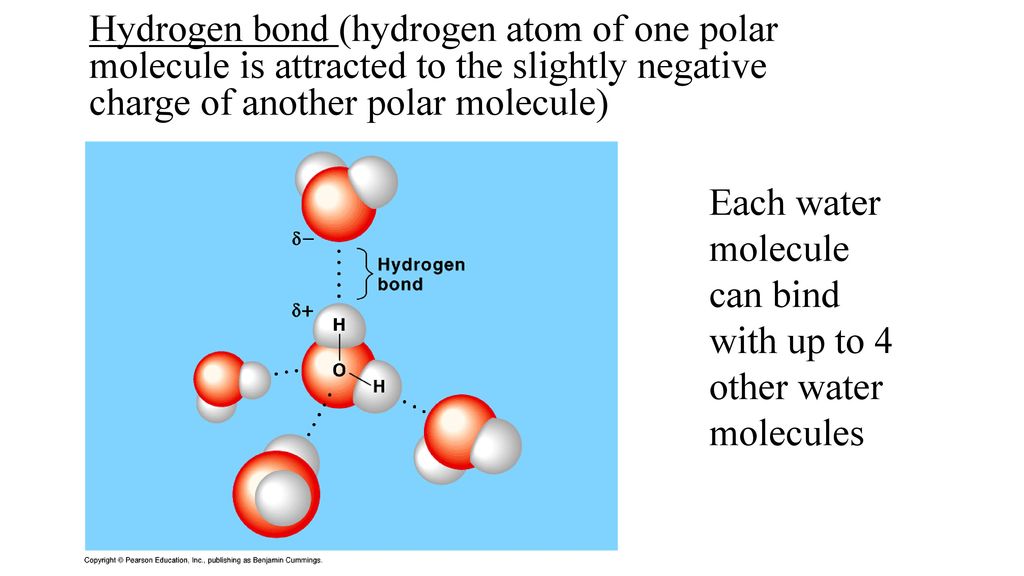
Think About Water 1.How many water molecules are present? (18) 2.How many Hydrogen bonds have formed between water molecules? (15) 3.Why can hydrogen bonds. - ppt download
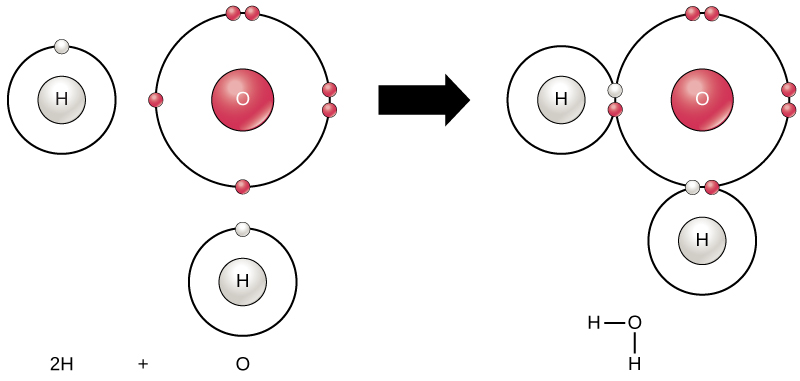
Both water and methanol have anomalously high boiling points due to hydrogen bonding, but the boiling point of water is greater than that of methanol despite its lower molecular mass. Why? -