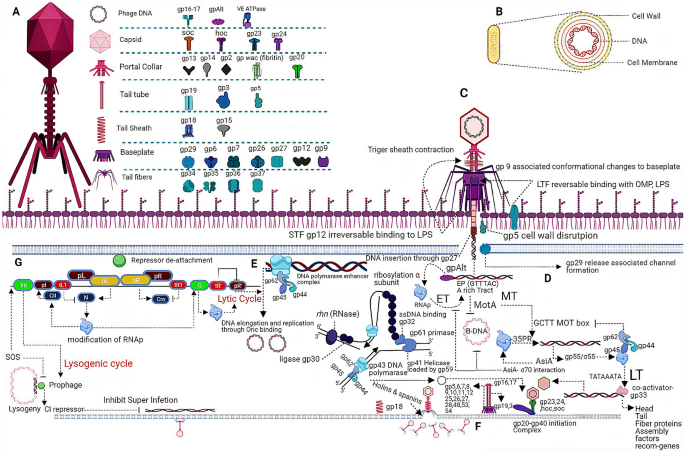
Structural and functional characterization of the receptor binding proteins of Escherichia coli O157 phages EP75 and EP335 - ScienceDirect

Phage–antibiotic combinations: a promising approach to constrain resistance evolution in bacteria - North - 2021 - Annals of the New York Academy of Sciences - Wiley Online Library

The Presence of Two Receptor-Binding Proteins Contributes to the Wide Host Range of Staphylococcal Twort-Like Phages | Applied and Environmental Microbiology
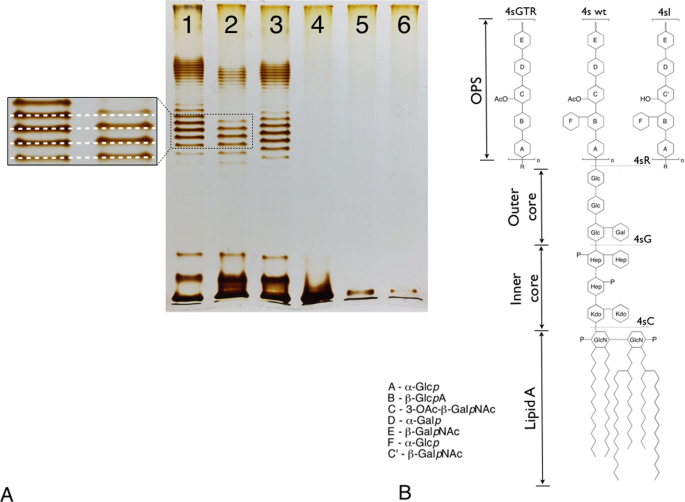
High-throughput LPS profiling as a tool for revealing of bacteriophage infection strategies | Scientific Reports

More than Rotating Flagella: Lipopolysaccharide as a Secondary Receptor for Flagellotropic Phage 7-7-1 | Journal of Bacteriology
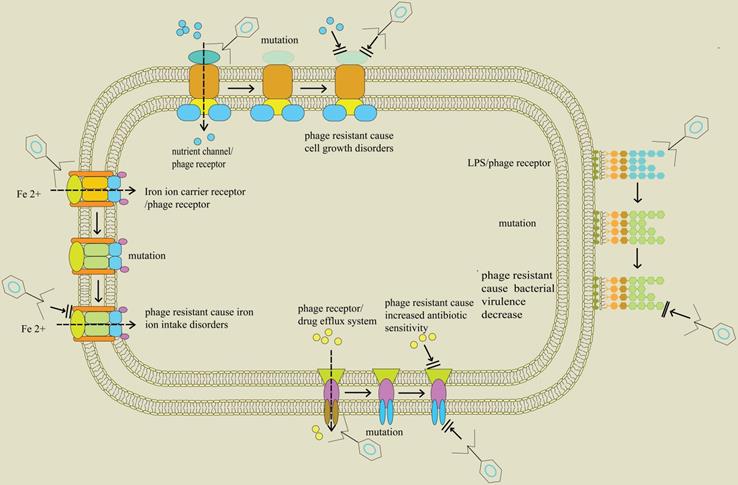
Receptor Diversity and Host Interaction of Bacteriophages Infecting Salmonella enterica Serovar Typhimurium | PLOS ONE

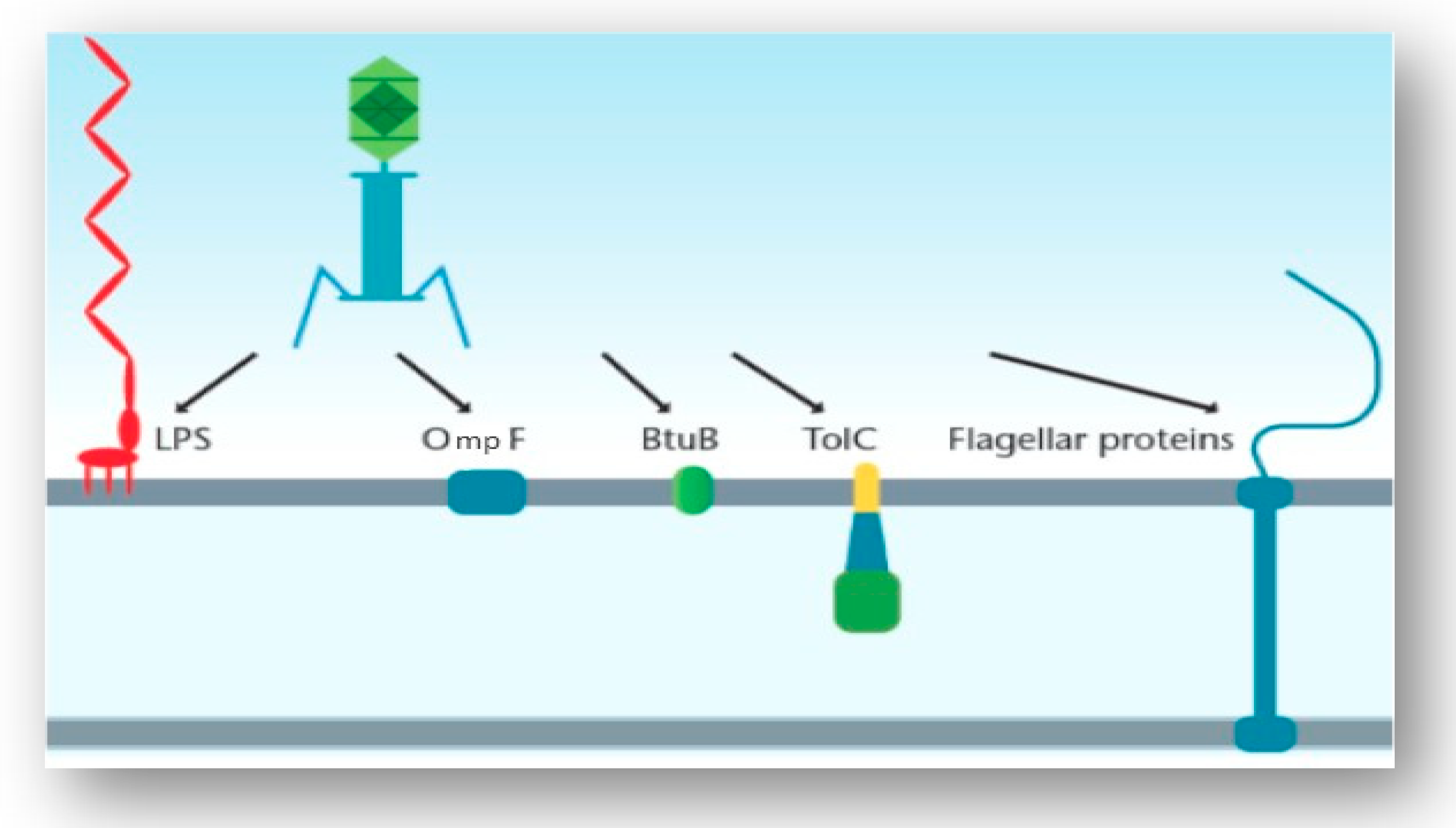
Examples of the possible host cell receptors a tailed phage generally... | Download Scientific Diagram

Exploitation of a Klebsiella Bacteriophage Receptor-Binding Protein as a Superior Biorecognition Molecule | ACS Infectious Diseases

A lipopolysaccharide-dependent phage infects a pseudomonad phytopathogen and can evolve to evade phage resistance | bioRxiv
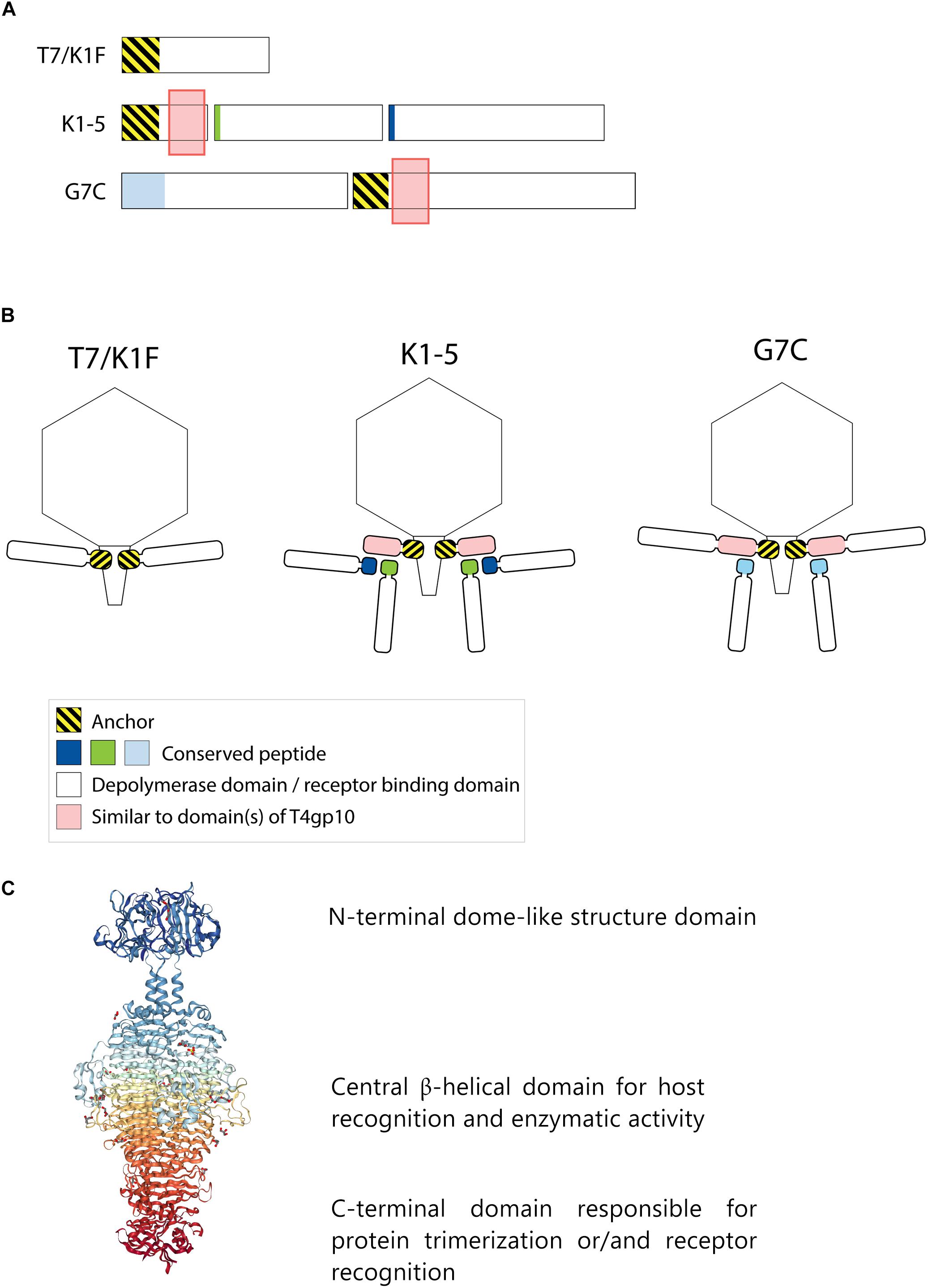
Frontiers | Modeling the Architecture of Depolymerase-Containing Receptor Binding Proteins in Klebsiella Phages

Novel Host Recognition Mechanism of the K1 Capsule-Specific Phage of Escherichia coli: Capsular Polysaccharide as the First Receptor and Lipopolysaccharide as the Secondary Receptor | Journal of Virology

High-throughput discovery of phage receptors using transposon insertion sequencing of bacteria | PNAS

Pseudotyping Bacteriophage P2 Tail Fibers to Extend the Host Range for Biomedical Applications | ACS Synthetic Biology

Systematic Discovery of Salmonella Phage-Host Interactions via High-Throughput Genome-Wide Screens | bioRxiv

Pseudotyping Bacteriophage P2 Tail Fibers to Extend the Host Range for Biomedical Applications | ACS Synthetic Biology
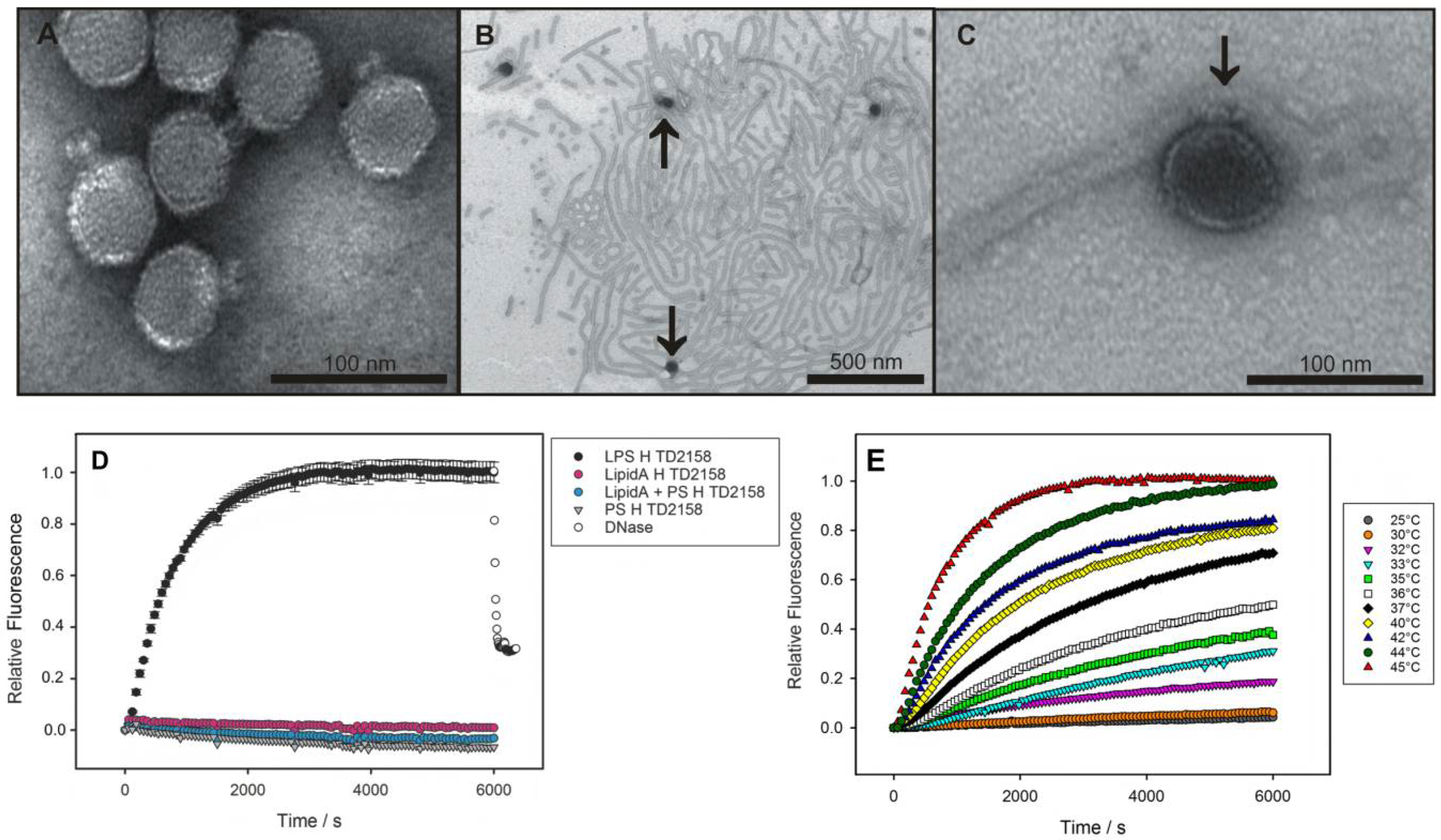
Viruses | Free Full-Text | In Vitro Studies of Lipopolysaccharide-Mediated DNA Release of Podovirus HK620

Not a barrier but a key: How bacteriophages exploit host's O‐antigen as an essential receptor to initiate infection - Broeker - 2017 - Molecular Microbiology - Wiley Online Library