Aqueous‐Phase Selective Oxidation of Methane with Oxygen over Iron Salts and Pd/C in the Presence of Hydrogen - Kang - 2019 - ChemCatChem - Wiley Online Library

KINETICS OF THE Fe(III) INITIATED DECOMPOSITION OF HYDROGEN PEROXIDE: EXPERIMENTAL AND MODEL RESULTS | Semantic Scholar

fenton reagent reaction hydrogen peroxide iron sulfate | Fundamental Photographs - The Art of Science

Oxidation of Wine Phenolics: A Critical Evaluation and Hypotheses | American Journal of Enology and Viticulture

Potentiation of hydrogen peroxide toxicity: From catalase inhibition to stable DNA-iron complexes - ScienceDirect

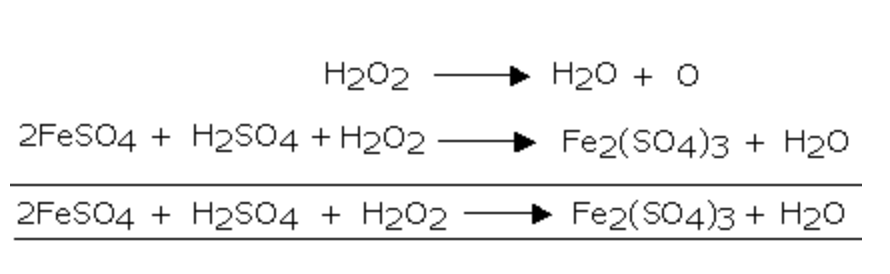
Objectives Define oxidation and reduction in terms of electron loss and gain. Deduce the oxidation number of an element in a compound. State the names. - ppt download
![PDF] Decomposition of hydrogen peroxide - kinetics and review of chosen catalysts | Semantic Scholar PDF] Decomposition of hydrogen peroxide - kinetics and review of chosen catalysts | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f2948b1ccb4f9f878523a741f22c7a299fa747b2/2-Figure1-1.png)
PDF] Decomposition of hydrogen peroxide - kinetics and review of chosen catalysts | Semantic Scholar

Activation of Hydrogen Peroxide by a Titanium Oxide-Supported Iron Catalyst: Evidence for Surface Fe(IV) and Its Selectivity | Environmental Science & Technology
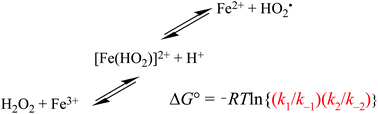
The principle of detailed balancing, the iron-catalyzed disproportionation of hydrogen peroxide, and the Fenton reaction - Dalton Transactions (RSC Publishing)

Objectives Define oxidation and reduction in terms of electron loss and gain. Deduce the oxidation number of an element in a compound. State the names. - ppt download